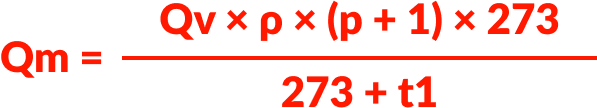Calculation method for heating a circulating fluid
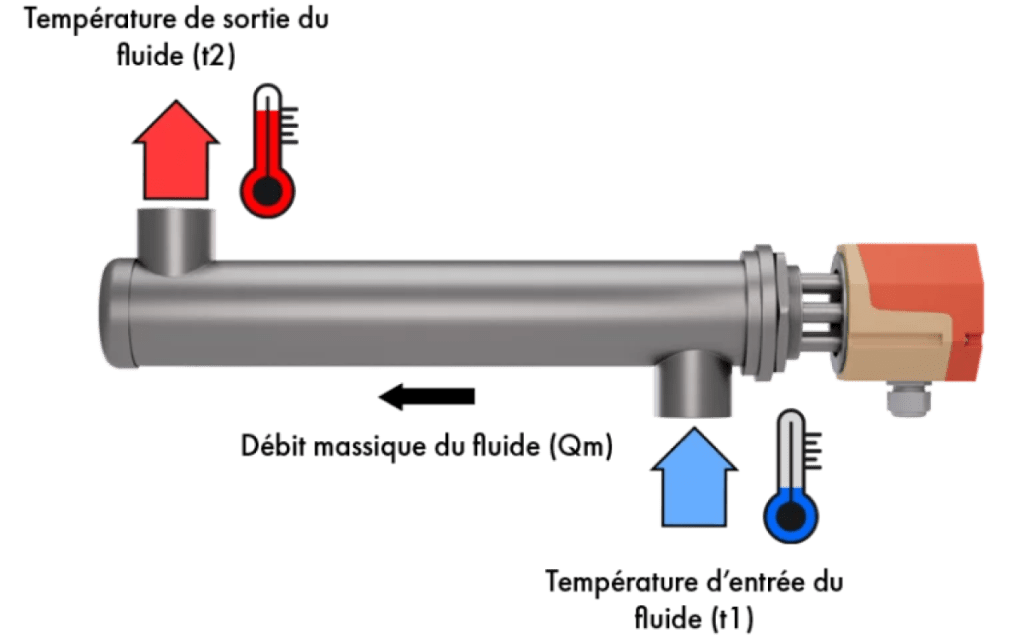
Calculating the heating power required to heat a circulating fluid
How do you calculate the power required to raise the temperature of a fluid (liquid or gas), in a single pass, circulating continuously in a heater or air-heating coil?
The temperature difference between inlet and outlet is guaranteed to be established after several minutes of operation. On start-up, all the equipment - the heater body, coil casing and resistor tube - are at ambient temperature. It is therefore not possible to instantly obtain the desired fluid outlet temperature.
This calculation is not applicable when the fluid is heated progressively by successive passes through the heater.
Calculating the power required to heat a circulating fluid
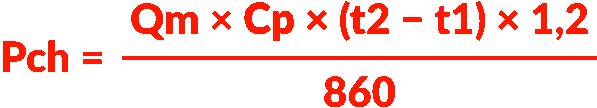
List of values for the heating capacity calculation formula
- Heating capacity: Pch (kW)
- Mass flow rate: Qm (kg/h)
- Specific fluid heat: Cp (kcal/kg .°C)
- Inlet temperature: t1 (°C)
- Desired output temperature: t2 (°C)
- Safety factor: 1.2
Calculating mass flow for a liquid (Qm)
List of values for calculating the mass emission of a liquid
- Mass flow rate: Qm (kg/h)
- Volume flow: Qv (dm3/h or l/h)
- Liquid density: ρ (kg/dm3)
p-values for some liquids
- Water: 1
- Mineral oil: 0.9
- Bitumen: 1.1
- Acetic acid: 1.1
- Hydrochloric acid: 1.2
- Nitric acid: 1.5
Calculating mass flow for a gas (Qm)
Volume flow given in Nm3/h
List of values for calculating the mass flow of a gas with volume flow in Nm3/h
- Mass flow rate: Qm (kg/h)
- Volume flow: Qv (Nm3/h)
- Gas density: ρ (kg/m3)
Volume flow given in m3/h
List of values for calculating the mass flow of a gas with volume flow in m3/h
- Mass flow rate: Qm (kg/h)
- Volume flow: Qv (m3/h)
- Operating pressure at heater inlet: p (bar.g)
- Gas inlet temperature: t1 (°C)
- Gas density: ρ (kg/m3)
Density values ρ for some gases (kg/m3)
at atmospheric pressure and 0°C
- Acetylene: 1.17
- Dry air: 1.29
- Ammonia: 0.77
- Nitrogen: 1.25
- Chlorine: 3.2
- Natural gas: 0.75
- Carbon dioxide: 2
- Hydrogen: 0.09
- Oxygen: 1.42
- Water vapour: 0.59
Specific heat (Cp)
Specific heat is the quantity of heat required to raise the temperature of one kilogram of a substance by one degree Celsius. It varies from material to material, reflecting their capacity to store or release thermal energy.
Cp values for some liquids (kcal/kg.°C)
- Water: 1
- Mineral oil: 0.5
- Bitumen: 0.58
- Acetic acid: 0.51
- Hydrochloric acid: 0.6
- Nitric acid: 0.66
Cp values for some gases (kcal/kg.°C)
- Acetylene: 0.40
- Dry air: 0.24
- Ammonia: 0.48
- Nitrogen: 0.25
- Chlorine: 0.12
- Natural gas: 0.50
- Carbon dioxide: 0.20
- Hydrogen: 3.4
- Oxygen: 0.22
- Water vapour: 0.49